Health Library
Retatrutide Side Effects: What Clinical Trials Tell Us So Far
February 15, 2026
Question on this topic? Get an instant answer from August.
The most common side effects of retatrutide are digestive issues like nausea, diarrhea, vomiting, and constipation. These are similar to what people experience with other GLP-1 medications like semaglutide and tirzepatide. They tend to show up early in treatment, especially during dose increases, and most people find they ease up as the body adjusts.
Retatrutide still an investigational drug. It has not been approved by the FDA yet and is currently in Phase 3 clinical trials. So everything we know about its side effects comes from trial data. But there is a meaningful amount of data to look at, including results from the recent TRIUMPH-4 Phase 3 trial.
What Are Most Common Side Effects?
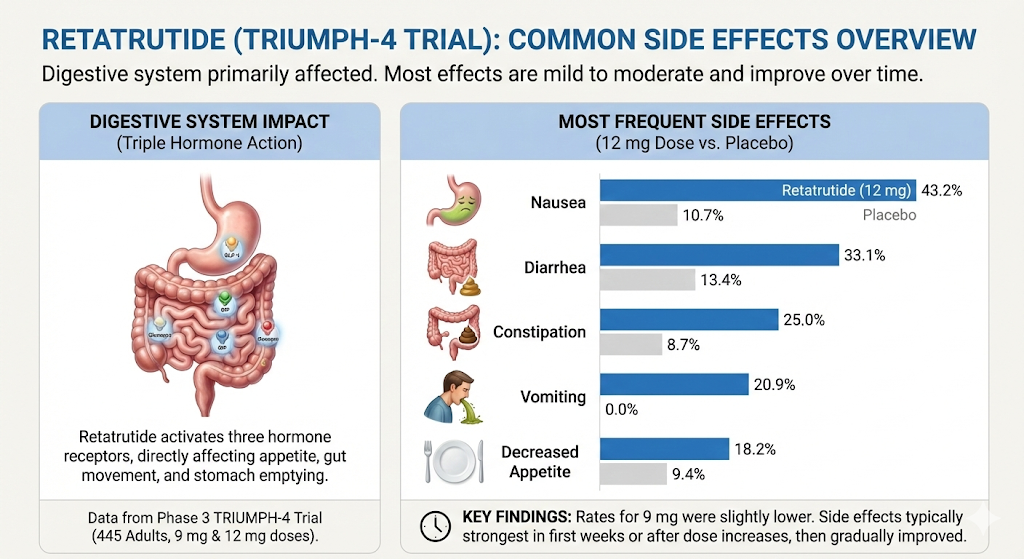
The digestive system takes biggest hit, at least in beginning. This makes sense because retatrutide works by activating three hormone receptors (GLP-1, GIP, and glucagon) that directly affect appetite, gut movement, and how quickly your stomach empties.
In Phase 3 TRIUMPH-4 trial, which tested two highest doses (9 mg and 12 mg) in 445 adults, most frequently reported side effects at 12 mg dose compared to placebo were:
- Nausea (43.2% vs 10.7% with placebo)
- Diarrhea (33.1% vs 13.4% with placebo)
- Constipation (25.0% vs 8.7% with placebo)
- Vomiting (20.9% vs 0.0% with placebo)
- Decreased appetite (18.2% vs 9.4% with placebo)
The 9 mg dose showed slightly lower rates across board. These numbers are higher than what you typically see with semaglutide or tirzepatide, which makes sense given that retatrutide is a more potent triple-action drug. But pattern is familiar. These are same types of side effects that come with any medication in this class.
The important thing to know that most of these side effects were rated as mild to moderate. They were most noticeable during first weeks of treatment and during dose increases, then gradually improved as participants stayed on a stable dose.
Does Retatrutide Affect Heart Rate?
This something researchers have been watching closely. In Phase 2 trials, some participants on higher doses had a small increase in resting heart rate, averaging about 5 to 7 extra beats per minute. This peaked around week 24 and then gradually came back down by weeks 36 to 48.
A small number of participants also experienced mild heart rhythm changes. However, no increase in heart attacks or serious cardiovascular events has been reported in any retatrutide trial so far. In fact, participants actually showed improvements in blood pressure, cholesterol, and other cardiovascular markers. In TRIUMPH-4 trial, 12 mg dose reduced systolic blood pressure by an average of 14 mmHg.
So while slight heart rate bump is worth monitoring, the overall cardiovascular picture looks encouraging based on what we know today.
What Is Dysesthesia Signal?
This is a newer finding that came out of TRIUMPH-4 Phase 3 results. Dysesthesia is an unusual or unpleasant sensation on skin, sometimes described as tingling, numbness, or a burning feeling. It was reported in 8.8% of participants on 9 mg dose and 20.9% on 12 mg dose, compared to just 0.7% with placebo.
This side effect was not seen in earlier Phase 2 trials, so it is something new that analysts and researchers are paying attention to. Eli Lilly described cases as generally mild, and it rarely led people to stop treatment. But with seven more Phase 3 trial readouts expected in 2026, this will be closely tracked to understand how common it really is and whether it resolves over time.
Are There Serious Side Effects?
Serious adverse events with retatrutide have been rare in trials so far. But a few possibilities are on radar because they have been seen with other GLP-1 type medications.
Here is what researchers are monitoring:
- Pancreatitis (inflammation of pancreas) has been reported rarely with GLP-1 drugs as a class. No confirmed cases have been specifically tied to retatrutide, but it remains something doctors watch for. Warning signs include severe, persistent abdominal pain that may radiate to back.
- Gallbladder problems, including gallstones, have been associated with rapid weight loss and with other incretin based medications. Trials are tracking this with retatrutide as well.
- Hypoglycemia (dangerously low blood sugar) was not a major issue in trials. No cases of severe hypoglycemia were reported, which is reassuring.
- Liver and kidney function were monitored throughout trials, and no significant safety signals emerged in these areas.
It is worth noting that no fatalities were linked to retatrutide in any clinical trial data published to date.
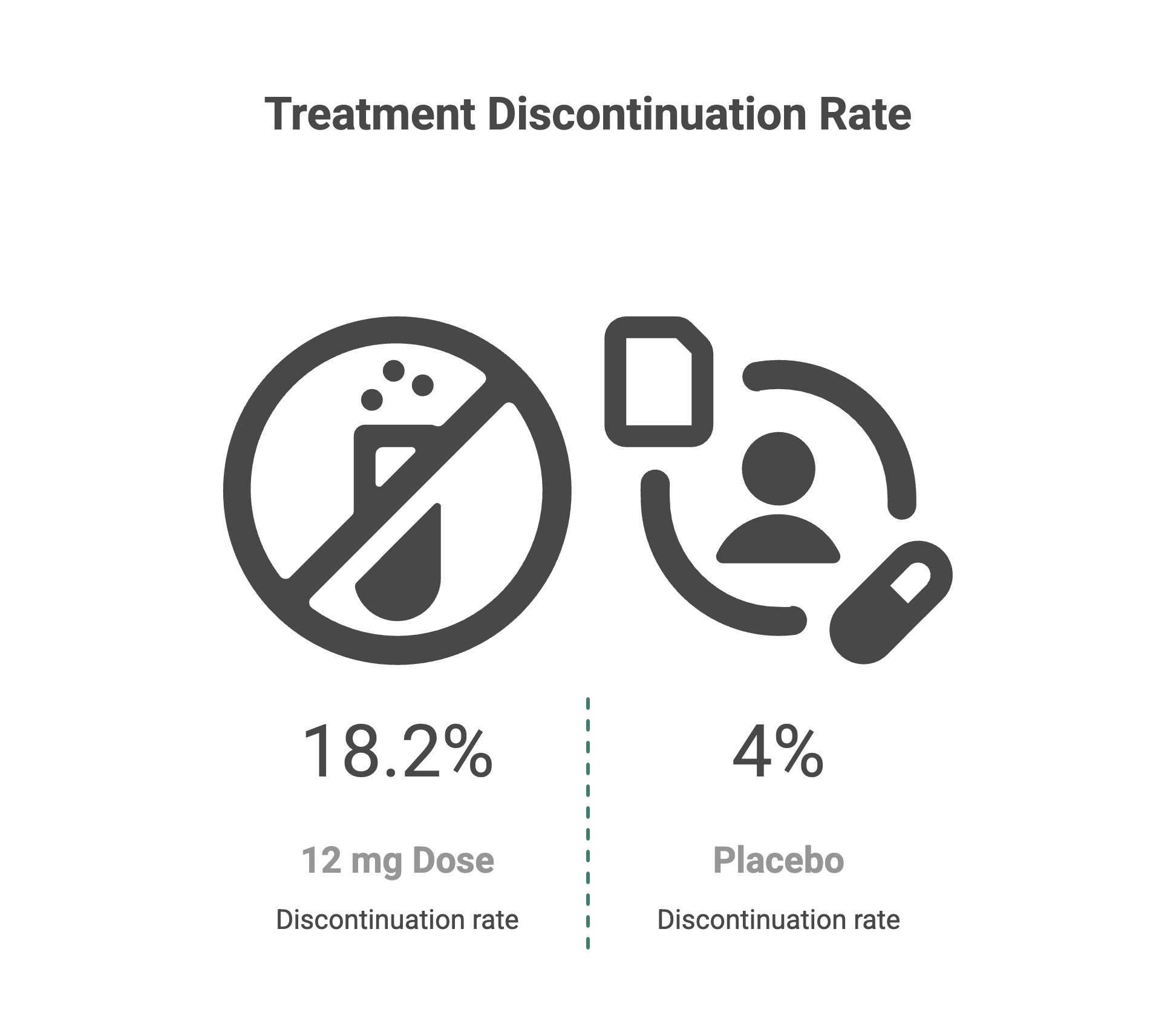
How Many People Stopped Treatment Because of Side Effects?
In TRIUMPH-4 trial, discontinuation rates due to adverse events were 12.2% at 9 mg dose and 18.2% at 12 mg dose, compared to 4% with placebo. Some of those discontinuations were actually because of what Lilly described as "perceived excessive weight loss," not because of feeling unwell.
When researchers looked only at participants with a BMI of 35 or higher at baseline (meaning those with more significant obesity), discontinuation rates dropped to 8.8% and 12.1% for two doses. This suggests that people with a higher starting weight may tolerate drug better, possibly because weight loss pace feels less extreme for them.
Why Does Gradual Dosing Matter?
Retatrutide trials use a stepwise dosing approach for a reason. Participants start at a low dose of 2 mg and increase every four weeks until they reach their target dose. This slow climb gives your body time to adjust to each level before moving to next one.
This approach is single most effective way to reduce side effects. Jumping straight to a high dose would make nausea, vomiting, and other digestive symptoms significantly worse. The gradual increase is also why most participants found that their early side effects faded within the first 8 to 12 weeks of reaching a stable dose.
What Do We Still Not Know?
Because retatrutide has only been studied for up to 68 weeks in its longest trials, long-term safety data beyond one year still limited. We do not yet know how the side effect profile looks over two, five, or ten years of use. Questions about potential effects on hair loss, bone density, muscle mass, and thyroid health remain open until longer studies provide answers.
Eli Lilly has seven additional Phase 3 readouts expected in 2026, covering obesity, type 2 diabetes, sleep apnea, liver disease, chronic back pain, and cardiovascular outcomes. These studies will paint a much fuller picture of both the benefits and the risks.
If retatrutide eventually earns FDA approval, your doctor will be able to walk you through whether the side effect profile makes sense for your situation. For now, the data shows a drug with powerful weight loss results and a side effect profile that manageable for most people, especially with careful, gradual dosing.
Health Companion
trusted by
6Mpeople
Get clear medical guidance
on symptoms, medications, and lab reports.